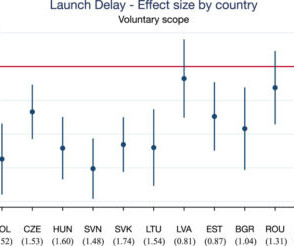
EMA and delays in drug launch
Healthcare ECONOMIST
APRIL 8, 2024
If the EMA evaluates a pharmaceutical and grants it marketing authorization, this determination is binding all all European Union member states. This procedure was adopted in 2001, and includes new treatments that are outside of the CP such as other pharmaceuticals and generics. Centralized procedure (CP).






































Let's personalize your content