Relyvrio Approval: Lessons Learned
Health Advances
OCTOBER 31, 2022
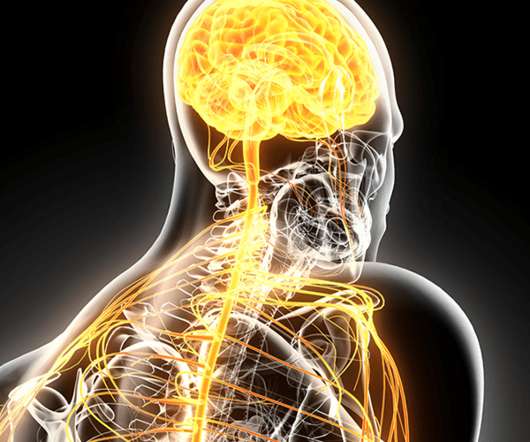
Together, these approvals show FDA is willing to be flexible and approve products based on more modest data packages that rely on limited phase II data, post-hoc analyses, and/or surrogate endpoints for neurodegenerative diseases with large unmet need.




Let's personalize your content